Conventional water-softening devices uses an ion-exchange resin in which “hardness” ions trade places with sodium ions that are electrostatically bound to the anionic functional groups of the polymeric resin. A class of minerals called zeolites also exhibits ion-exchange properties; these minerals were widely used in earlier water softeners. Water softeners may be desirable when the source of water is a well, whether municipal or private.
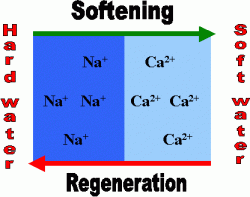
The water to be treated passes through a bed of the resin. Negatively-charged resins absorb and bind metal ions, which are positively charged. The resins initially contain univalent hydrogen, sodium or potassium ions, which exchange with divalent calcium and magnesium ions in the water. As the water passes through the resin column, the hardness ions replace the hydrogen, sodium or potassium ions which are released into the water. The “harder” the water, the more hydrogen, sodium or potassium ions are released from the resin and into the water. Resins are also available to remove carbonate, bi-carbonate and sulphate ions which are absorbed and hydroxyl ions released from the resin. Both types of resin may be provided in a single water softener.
Regeneration:
As these resins become loaded with undesirable cations and anions they gradually lose their effectiveness and must be regenerated. If a cationic resin is used (to remove Calcium and Magnesium ions) then regeneration is usually effected by passing concentrated brine, usually of sodium chloride or potassium chloride, or hydrochloric acid solution through them. For anionic resins a solution of sodium or potassium hydroxide (Lye) is used. Most of the salts used for regeneration gets flushed out of the system and may be released into the soil or sewer. These processes can be damaging to the environment, especially in arid regions. Some jurisdictions prohibit such release and require users to dispose of the spent brine at an approved site or to use a commercial service company. Most water softener manufacturers provide metered control valves to minimize the frequency of regeneration. It is also possible on most units to adjust the amount of reagent used for each regeneration. Both of these steps are recommended to minimize the impact of water softeners on the environment and conserve on reagent use. Using acid to regenerate lowers the pH of the regeneration waste.
In industrial scale water softening plants, the effluent flow from re-generation process can be very significant. Under certain conditions, such as when the effluent is discharged in admixture with domestic sewage, the calcium and magnesium salts may precipitate out as hardness scale on the inside of the discharge pipe.
If Potassium chloride is used the same exchange process takes place except that potassium is exchanged for the calcium, magnesium and iron instead of sodium. This is a more expensive option and may be unsuited for people that are on potassium-restricted diets.